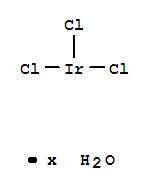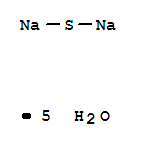
1313-83-3
- Product Name:SODIUM SULFIDE PENTAHYDRATE
- Molecular Formula:H2O . 1/5 Na2 S
- Purity:99%
- Molecular Weight:168.12
Product Details;
CasNo: 1313-83-3
Molecular Formula: H2O . 1/5 Na2 S
Quality Manufacturer Supply High Purity SODIUM SULFIDE PENTAHYDRATE 1313-83-3 Low Price
- Molecular Formula:H2O . 1/5 Na2 S
- Molecular Weight:168.12
- Melting Point:120℃ [MER06]
- PSA:71.45000
- Density:1.580
- LogP:-0.85690
SODIUM SULFIDE PENTAHYDRATE(Cas 1313-83-3) Usage
|
Description |
Sodium sulfide pentahydrate, represented as Na2S·5H2O, is a colorless and hygroscopic solid compound derived from sodium carbonate and a sulfidizing gas mixture. It possesses a density of 1.58 g/cm3. This sulfide salt, belonging to the inorganic sodium salt category, finds applications in diverse industries, including pulp and paper, water treatment, textiles, and various chemical manufacturing processes. In addition to its industrial uses, sodium sulfide pentahydrate has a unique application in gel form for softening toenails, aiding in trimming, and providing pain relief for ingrown toenails. |
| Notes | However, caution is warranted in handling sodium sulfide, as it is corrosive to the eyes, skin, and respiratory tract. Exposure may lead to skin allergies, and the compound has the potential to explode in air. |
|
Chemical Properties |
flat, shiny crystal(s); flammable; evolves H2S in acid solutions [MER06] [ALD94] |
InChI:InChI=1/Na.5H2O.H2S/h;6*1H2/q+1;;;;;;
1313-83-3 Relevant articles
The Synthesis of Sodium Sulfide Pentahydrate, Na2S·5H2O, Through a Solid-Gas Reaction of Sulfidizing Gas Mixture with Sodium Carbonate, Na2CO3
Halil Güler,Figen Kurtuluş,İmdat Kadan,Ayşe Morkan &Sebahat Akin
Phosphorus, Sulfur, and Silicon and the Related Elements Volume 181, 2006 - Issue 6
Sodium sulfide pentahydrate, Na2S·5H2O, was prepared from sodium carbonate, Na2CO3, via a sulfidizing gas mixture by a solid-gas reaction under the cooling of a nitrogen atmosphere. The observed phase was found to be the pentahydrated form of sodium sulfide.
Relevant Products
-
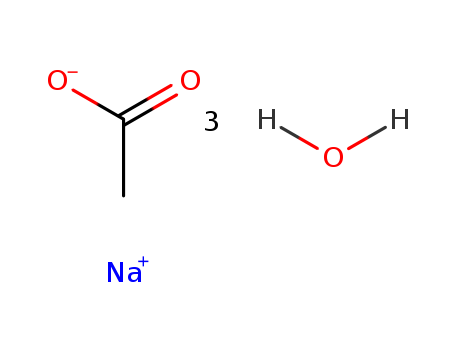
Sodium acetate trihydrate
CAS:6131-90-4
-
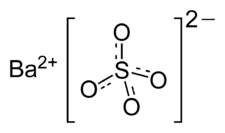
Barium sulfate
CAS:7727-43-7
-
Iridium(III) chloride hydrate
CAS:14996-61-3